In spite of the great advances in the diagnosis and treatment of malignant tumors, cancer continues be one of the principal causes of death in the highly industrialized countries. It is calculated that one out of four persons will eventually die from some form of cancer. Since it is true that surgery and radiotherapy are capable of curing some patients with localized tumors and that chemotherapy has achieved cures in some ten types of malignant tumors, the general mortality rate from cancer has not improved substantially in the last 25 years and nearly 60 percent of the patients, upon being diagnosed, find that their disease is so widespread that the chemotherapy drugs currently being used, due to their high toxicity, cannot be given in dosages sufficient to destroy the large tumoral mass present in patients. Many cannot be exposed to therapy, surgery or radiotherapy because of the undesirable effects. There are several types of tumors for which there is no effective treatment yet known.
All this justifies, and even makes imperative, the search for new substances with anti-tumor effect and ideally, with little or no toxicity in therapeutic doses. In the last ten years, several vegetable and hormonal substances have been discovered with such characteristics and, therefore, many patients who formerly could not be benefited or alleviated medicinally may now be exposed to useful , antineoplastic treatments.
KEM S.A. Laboratories, with great satisfaction, is able to present a vegetable agent whose anti-tumor action was known empirically for many years, but in the last twenty years has been scientifically proven, primarily through the clinical studies directed by Dr. Ernesto Contreras Rodriguez and carried out in the Centro Medico y Hospital Del Mar at Playas de Tijuana, B.C.N. Mexico.
This anti-tumor agent is AMYGDALIN (commonly known as Laetrile).
AMYGDALIN is a natural substance that can be found in a variety of species in the vegetable kingdom. The greatest concentration is found in the seeds of the rosaceous fruits, such as the apricot pits and other biter nuts. There are many seeds, cereals and vegetables that contain minimal quantities of Amygdalin and form part of our daily diet.
Various documents from the oldest civilizations such as Egypt at the time of the Pharaohs and from China 2,500 years before Christ mention the therapeutic use of derivatives of bitter almonds. Egyptian papyri from 5,000 years ago mention the use of "aqua amigdalorum" for the treatment of some tumors of the skin. The Greeks and Romans also attributed therapeutic properties to that extract in low doses. But the systematized study of AMYGDALIN really did not begin until the first half of the past century, when the chemist Bohn discovered in 1802 that during the distillation of the water from bitter almonds hydrocyanic acid was released.
Soon many researchers became interested in analyzing this extract and so Robiquet and Boutron isolated, for the first time, a white crystalline substance which they called AMYGDALIN (from amygdala = almond)
Leiberg and Wholler in 1937 isolated an enzymatic compound from sweet almonds, also present in the biter ones, which they called emulsin. They later reported that emulsin broke AMYGDALIN down into three compounds: glucose, hydrocyanic acid, and benzaldehyde.
Studies from that time, performed by several authors, can summarize the declaration made by Otto Jacobsen in his book "Die Glucoside" in 1887: "AMYGDALIN is not toxic," and gives 99 references from studies made within the 20 years prior to his publication.
Extraction Methods
The source for obtaining AMYGDALIN which use most currently utilized use the apricot kernel. The method for its extraction and refining has only been standardized in the last five years, thanks to the multiple research projects completed primarily by KEM S.A. Laboratories. The main steps to follow in the extraction of AMYGDALIN are summarized below :
1. Selection of kernels.
2. Cleaning the Kernels. Keeping
as the principal objective the elimination of shells, husks, rock and leaves
which might damage the mills.
3. Milling the kernels. With
this, it is possible to separate most of the oil from the initial milling
and the remainder in the subsequent millings. The product from the milling
is a fine, brownish powder with a bitter odor which, upon letting it sit,
ferments spontaneously, which is desirable, because with it the yield from
the extraction is increased.
4. Dissolving the powder. A stainless
steel digester is used in the presence of isopropyl alcohol at a constant
temperature and with constant reflux of the alcohol, for a period of 2
1/2 hours.
5. Filtering. One uses filters
on very fine cloth so as to avoid the passage of particles of the powder
which are not digested. The resulting product is allowed to sit for 72
hours, and is then washed with ethylic ether eliminate the isopropyl alcohol.
6. Drying the AMYGDALIN. The
dry, white powder, product of the filtering, is spread out on stainless
steel dishes and is exposed for eight hours to temperatures between 50
and 60 degrees C. The project is 65 through 80% percent pure AMYGDALIN.
7. Crystallization. This is completed
in closed, glass, vacuum receptacles after exposing the AMYGDALIN to a
mixture of activated carbon and ionized earth which act as a filter. The
final product is AMYGDALIN with a purity of no less than 96%.
Physical and chemical properties
Although the identification of the majority of the physical and chemical characteristics of AMYGDALIN have been known since the beginning of our century, it was not until the second half of this century that Ernest T. Krebs Jr. (biochemist) and Ernest T. Krebs Sr. (doctor) isolated Amygdalin with a purity of practically 100%, enabling all the physical and chemical characteristics peculiar to AMYGDALIN to be ascertained. Listed below are the majority of these:
1. Amygdalin is a white, crystalline, inodorous powder with an intensely bitter taste, slightly soluble in cold water, alcohol and acetone, very soluble in hot water, alcohol and acetone. Insoluble in ether.
2. It has a pH of 7 (neutral) in a saturated, aqueous solution. its point of fusion is between 210 and 218 degrees Celsius and its loss upon drying is less than 12%.
3. Its optical rotation is levogyrous or negative: between -37 and 42 degrees. It has a maximum absorbency of ultraviolet light of 262 microns and a minimum of 250 microns.
4. Its stability is complete in crystalline form as well as in saturated , aqueous solution in which the loss is less than 2.5% after 5 years.
5. Chemically, it is cyanogenic diglucoside, with a condensed formula C20H27NO11, with a molecular weight of 457.42 g., a chemical name of D (1) Mandelonetrile-betaglucoside-6 beta-D-glucoside.
6. If it is mixed with concentrated, hydrochloric acid, it gives positive reactions characteristic of benzaldehyde, of the reducing sugars and the hydrocyanic acid.
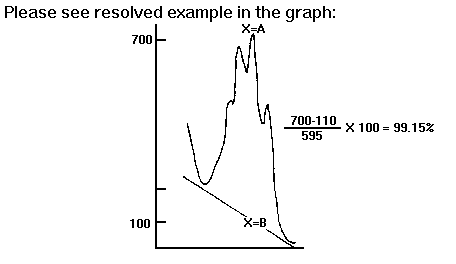
Evaluation Test
It is considered to be specific and, therefore, useful in determining the purity of the AMYGDALIN product.
Method
1. Measure out a volume equivalent to 100mg of AMYGDALIN and transfer
it to a volumetric matrass.
2. Add distilled water to complete 100 ml. solution.
3. Measure the absorbency of the solution in ultraviolet light at
230 u and up to 280 u, using a spectrophotometer with a quartz cell of
1 cm.
4. Graph the curve of the absorbency of the solution in ultraviolet
light.
5. Determine the tangent between the minimum absorbency (near 230
u) and the final inflection (at 280 u).
6. Designate at point A the maximum absorbency in the curve, and
at point B the corresponding absorbency of the traced tangent.
7. Formula and calculation. A constant equivalent to 595 will be
utilized as divisor, and the following operation will be performed:
A-B x 100 = PERCENTAGE OF AMYGDALIN IN SAMPLE 595
 |
PRECLINICAL STUDIES WITH AMYGDALIN
It is accepted that every scientific study of a substance that is intended for human use should include toxicology studies with laboratory animals, in order to predict safe dosages for humans in a more rapid and rational manner than if it id calculated solely in an empirical fashion. Generally, toxicology studies should include the determination of the LD50 (lethal dosage for 50% of those exposed) in mice and other mammiferous rodents, specially by intravenous, intraperitoneal, intramuscular, subcutaneous and occasionally tumorally or intra-cavity applications if the product is intended for use in this form. It is also indispensable to calculate the Minimum Toxic Dosage and the Minimum Lethal dosage in rodents as well as in dogs, in those in which also should be studied the histopathological changes that the exposure to the substance caused in at least six organs considered to be vital. Oral toxicity is also studied in dogs.
Since it is true that there is no animal that reacts to medicaments exactly like man, it is know that by using a rodent, or a dog, in the toxicology tests, very likely one can predict the majority of the pharmacological effects of said substance in a human being. There are few cases in which it is better to use monkeys rather than dogs for these studies. AMYGDALIN has been subject to these studies under the sponsorship of foreign investigators as well as KEM S.A. LABORATORIES.
We shall make mention of only the results in the following pages. Other studies not included in this summary include experiments for antigenicity in guinea pigs which were reported as negative (not antigenic); experiments for teratogenicity in rats and dogs in which there were found no alterations in the progeny which could be attributed to the use of AMYGDALIN; Studies of subacute toxicity from various application methods and in several rodents which show a very low toxicity orally and practically none by intravenous or intraperitoneal application.
LD50 for Various Species and By Different Methods of Application
 |
Acute Toxicity by Intravenous Application:
At dosages up to 3,000 mg/kg/B.W. (or 54,000mg total dosages), it was not possible to achieve the minimum lethal dosage. There was only slight arterial hypotension and hyposphygamia which normalized before concluding the 30 minute infusion. The monitored blood flow and cardiac contractility showed no changes.
Acute Toxicity Through I.V. Application:
The minimum lethal dose was not achieved in dosages up to 15,000 mg/kg/B.W. (or 120,000mg total dosages). The general symptoms were slight arterial hypotension and fleeting adipsia, which appeared after the minimum toxic dosage of 3,750 mg/kg/B.W.
Subacute Intravenous Toxicity:
There were no undesirable effects up to daily dosages of 1,000 mg/kg/B.W. for eight weeks.
Chronic Intravenous Toxicity:
Dosages up to 15 mg/kg/B.W. five days per week, for six consecutive months, did not cause clinical or histopathological alterations in necropsy studies of the exposed dogs.
Acute Oral Toxicity:
The minimal lethal dose was 14,318 mg/kg/B.W. and the minimum toxic dose was 400 mg/kg/B.W. The symptoms that were observed were apathy, anorexia, adipsia, and loss of hair. These symptoms disappeared in less than 48 hours in the dogs in which AMYGDALIN was discontinued. The dog that received the minimum lethal dosage developed convulsions and typical cyanide poisoning shock which evolved into coma and death by cardiac arrest.
Oral Toxicity:
By calculation, some authors suggested as the minimum lethal dosage 2,000 to 2,500 mg/kg/B.W.
All these findings coincide with that mentioned by Otto Jacobsen in 1887, Davidson in 1944, and Dr. Dean Burk (National Cancer Institute of the USA) in 1968. "AMYGDALIN is impressively atoxic (non-toxic) from the pharmacological point of view," and that "Non-hydrolyzed AMYGDALIN is less toxic than glucose." On the other hand, our studies proved that KEMDALIN in dosages of 100 to 120 mg/kg/B.W. (unusual dosage for humans) by intravenous application corresponds to a dosage of AMYGDALIN 30 to 40 times less than the minimum toxic dosage in dogs and that KEMDALIN in dosages of 20 to 40 mg/kg/B.W. orally, used in humans, is some 10 to 20 times less than the minimum toxic dosage, orally, in dogs.
ANTINEOPLASTIC ACTIVITY OF AMYGDALIN IN THE CULTIVATION OF TUMOR CELLS AND IN VARIOUS TUMOR MODELS IN ANIMALS
With the purpose of exploring the antineoplastic activity of AMYGDALIN, several pre-clinical studies have been completed. There have been some in which the results have been negative, but this does not invalidate for AMYGDALIN or for other substances analyzed by a similar method, the positive anti-tumor findings reported in this study. These are listed by author or responsible institution.
Pre-clinical Studies to Demonstrate the Antineoplastic Activity of AMYGDALIN
Mark Paulic:
A study sponsored by the American Lung Association of Indiana, USA. With pulmonary tumor cells kept in ascitic liquid of mice, AMYGDALIN caused a statistically, significant increase in the mice inoculated and exposed to AMYGDALIN.
SCIND Laboratories:
In two studies using Carcinoma Walker 256 in rats, a statistically significant increase in the survival of rats exposed to AMYGDALIN was proved.
Harold Manner:
Several studies of mice with spontaneous breast cancer, type C3H/HEJ, have shown a statistically significant increase in the life span of those exposed to AMYGDALIN with proteolytic enzymes and mega-doses of Vitamin-A.
K. Sugiura:
Sponsored by the Sloan-Kettering Institute of New York, USA. Studies with spontaneous CD8F1 and Swiss albino mice, breast tumors, showed in five separate studies that AMYGDALIN has clear, anti-tumor effect demonstrated by the regression of the primary tumors and the retardance caused in the appearance of pulmonary metastasis in the mice exposed to AMYGDALIN.
SCIND Laboratories:
Using cancer cells from human, mammary melanoma exposed to AMYGDALIN, it was proved by the polarographic method of oxygen consumption, inhibition in activity and respiration.
We believe that these studies adequately justify
the study of the toxicity, tolerance, and anti-tumor effect of AMYGDALIN
in humans.
CLINICAL EXPERIENCE WITH THE USE OF AMYGDALIN
Like many other substances, AMYGDALIN was initially employed empirically on patients with malignant tumors. Inozensov, a Russian doctor, used it with this purpose at the beginning of our century. Dr. Ernest T. Krebs Sr., Dr. Manuel Navarro and Dr. Ernesto Contreras Rodriguez and their collaborators have published their experiences since the 1950�s.
All agree that it is a characteristically harmless substance when administered intravenously under medical supervision and that orally, therapeutic dosages can be tolerated. On the other hand, they all report definite palliative and anti-tumor effects even in patients with cancer in terminal stages. Already mentioned are the great numbers of scientific studies which, in animals, have shown the antineoplastic effect of AMYGDALIN during the last 25 years. In this section, summaries of the results of the clinical studies performed by Dr. Ernesto Contreras Rodriguez and his associates at the Centro Medico y Hospital Del Mar, Playas de Tijuana, B.C.N., Mexico, are included.
These studies were sponsored by KEM S.A. LABORATORIES, taking into account that Dr. Contreras and his staff have the greatest experience with the use of AMYGDALIN in humans: Nearly 30,000 patients treated. Phase I, II and III studies were completed.
Phase I Study
These studies were conducted to determine the minimum toxic dosage in humans. Some 420 patients with cancer in advanced stages and 90 healthy volunteers were exposed to AMYGDALIN in intravenous dosages of up to 21g, or 2g orally, per day, tolerated perfectly without evidence of toxicity, acute or chronic (six month study). The palliative effect was apparent in those patients who were not able to tolerate any kind of conventional treatment.
Phase II Study
The Phase II studies were designed to demonstrate the antitumoral effect of AMYGDALIN. The files of 1,200 patients with advanced malignant neoplasms exposed to AMYGDALIN in varying dosages were reviewed. Intravenously and orally, AMYGDALIN demonstrated to have anti-tumor effects. Complete remissions, partial remissions and prolonged stabilizations (objective responses) were seen in almost 33% of the patients, who were no longer candidates for conventional treatment in more than 70% of the cases. Subjective responses (responses of the patient rather than the malignancy) were observed in more than 45% of the cases. There was improvement in the general condition of the patient, improvement in appetite and in diminishing pain. We should emphasize that practically 100% of the patients using morphine derivatives to control their pain were able to substitute these for non-narcotic analgesics in less than four weeks after the initial dose of AMYGDALIN.
Phase II and III studies were sponsored to demonstrate the therapeutic value of AMYGDALIN upon specific types of tumors.
Currently, Phase III studies have been completed on patients with inoperable lung cancer of all types (four studies on previously or simultaneously exposed to conventional treatments and AMYGDALIN and one study on virgin patients treated only with AMYGDALIN) and in patients with Glioblastoma Multiform. Studies will soon be completed on patients with breast cancer, prostate, stomach, pancreas, and other malignant tumors who are not candidates for conventional treatments.
The findings from one of the Phase III studies (patients with inoperable lung cancer) are included in this summary.
Phase III Study
This study included 257 patients with inoperable lung cancer, in its different histopathologic types and stages of diffusion. It was possible to prove that there was complete or partial remission from the disease in 12.45% of the patients and, at least, stabilization of the disease for a minimum of six months in 55.25% of the patients. It was concluded that more than half of the patients experienced subjective improvement.
Survival to twelve months and the median survival were better than those of the historic controls used for comparative analysis, both from the diagnosis and from the date of first treatment. When the results were compared with some of the better series reported using of radiotherapy and/or chemotherapy, the results with AMYGDALIN were as good, or better, according to the histopathologic type, stage and functional capacity (Karnofsky Performance Status). The survival to twelve months from the diagnosis was 55.25% (142 of 257 patients) and 34.63% from the first use of AMYGDALIN (89 of 257 patients). The median survival was 59.45 weeks from the diagnosis and 35.76% from the first dose of AMYGDALIN. Most other literature reports a median survival of 25 weeks for patients with inoperable lung cancer.
There was not one case reported with morbidity or mortality attributable to the use of AMYGDALIN. All this in contrast to the frequent side effects reported with the use of radiotherapy in high dosages and/or multiple chemotherapy, mortality of 3 to 4% and a significant or severe morbidity in more than 30% of the patients who frequently discontinue their treatment.
It was also proven that AMYGDALIN combines perfectly with conventional
therapy, helpfully influencing in the tolerance and response to it, chiefly
by diminishing the severity of the side effects caused by chemotherapy.
AMYGDALIN is proposed to be used:
1. As an alternative on those patients who
are not candidates for other methods of conventional treatment.
2. As a simultaneous treatment with conventional
therapy.
3. As a helpful treatment for those patients
in remission.
4. As one more palliative for cancer patients
in advanced stages.
Conclusion
With all that which has been previously exposed, we can conclude that AMYGDALIN has an anti-tumor effect, even in those patients in poor condition and/or with an extensively disseminated disease. Non-toxic dosages, whether alone or in combination with other methods of conventional treatment, are useful. At KEM S.A. Laboratories we believe that, with the studies already conducted by the Centro Medico and Hospital Del Mar, and the ones still pending, we shall soon see the greater value of AMYGDALIN as an antineoplastic agent alone or associated with other conventional cancer treatments.
AMYGDALIN (or Laetrile) as an antineoplastic agent is no longer a dream to be proven, but rather, a demonstrated reality with scientific evidence confirmed each time that it is prescribed.
1. U.S. Department of Health Education and Welfare. End
results in cancer. Report No. 4. Washington, D.C., Government Printing
Office. 1974.
2. American Cancer Society. Cancer Facts and Figures.
1975, 1979.
3. Holland, James. Frei III, Emil. Cancer Medicine. Lea
and Febiger. Philadelphia, 1974.
4. Symington, T., Carter, R.L. Scientific Foundations
of Oncology. William Heinemann Medical Books Publications, Chicago, 1976.
5. Clark, Radolph., Cumley, R.W., Year Book of Cancer,
Year Book Medical Publishers, Inc. 1976, 1979.
6. Horton, J., Hill, G.J., Clinical Oncology, W.B. Saunders
Co., Pa. 1976.
7. Macdonald, E.J., Epidemiologic aspects to current
concepts in cancer. JAMA 228:884-886. May 13, 1974.
8. Summa, H.M., Amygdalin. A Physiological active therapeutic
agent in malignancies. Krebsgechehen, a., Jossa-Arznei, Steinan, Germany,
1972.
9. Dioscorides. Treasure of Drugs. Basel 1:34, 1598.
10. Plinius. De Morbis Nulierum. Naturgesch. Leipzig
139., 1881.
11. Bohm. Allgen. Jour. Chemmie. 10:126, 1802.
12. Robiquet y Boutron. Extraccion de la Amygdalina.
Ann. Chem. 44: 325, 1830.
13. Lieberg y Wholer. Emulsina. A 22:1, 1937. A 22:46,
1938.
14. Martens. Jahrb. In. Ausl. Med., XVII:1, 1838.
15. Inozemzov, F. Two fungus like tumors healed without
operation, by oral Amygdalin Gazet Med. de Paris. 37. 1845.
16. Mc Naughton, A. Protocol on Laetrile. Presented by
Mc Naughton Found. IND . 6734, 1970.
17. Krebs, E., Gurchot, Ch., Biochemistry of Laetrile.
Science 104: 132, 1946.
18. Navarro, M. et al. Mechanism of action and therapeutic
effects of Laetrile in Cancer. J. Phillip. Med Assc. 33:620-627., 1957.
20. Burk, D., et al. Hyperthermy