

When a neutron passes near to a heavy nucleus, for example uranium-235 (U-235), the neutron may be captured by the nucleus and this may or may not be followed by fission. Capture involves the addition of the neutron to the uranium nucleus to form a new compound nucleus. A simple example is U-238 + n ==> U-239, which represents formation of the nucleus U-239. The new nucleus may decay into a different nuclide. In this example, U-239 becomes Np-239 after emission of a beta particle (electron). But in certain cases the initial capture is rapidly followed by the fission of the new nucleus. Whether fission takes place, and indeed whether capture occurs at all, depends on the velocity of the passing neutron and on the particular heavy nucleus involved.
The probability that fission or any another neutron-induced reaction will occur is described by the cross-section for that reaction. The cross-section may be imagined as an area surrounding the target nucleus and within which the incoming neutron must pass if the reaction is to take place. The fission and other cross sections increase greatly as the neutron velocity reduces. Hence in nuclei with an odd-number of neutrons, such as U-235, the fission cross-section becomes very large at thermal energies.
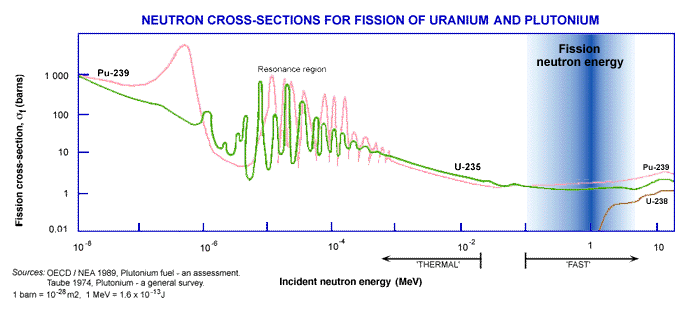
Note that both scales are logarithmic.
A neutron is said to have thermal energy when it has slowed down to be in thermal equilibrium with the surroundings (when the kinetic energy of the neutrons is similar to that possessed by the surrounding atoms due to their random thermal motion). Hence the main application of uranium fission is in thermal reactors fuelled by U-235 and incorporating a moderator such as water to slow the neutrons down. The most common examples of this are Light Water Reactors*.
*there are two main varieties, Pressurised Water Reactors and Boiling Water Reactors.
As implied previously, the fission cross-section is much reduced at high neutron energies (> 0.1 MeV) relative to its value at thermal energies. It is nonetheless possible to use this so-called fast fission in a fast reactor whose design minimises the moderation of the high-energy neutrons produced in the fission process.
* The chain reaction is started by inserting some beryllium mixed with polonium, radium or other alpha-emitter. Alpha particles from the decay cause the release of neutrons from the beryllium as it turns to carbon-12.
About 85% of the energy released is initially the kinetic energy of the fission fragments. However, in solid fuel they can only travel a microscopic distance, so their energy becomes converted into heat. The balance of the energy comes from gamma rays emitted during or immediately following the fission process and from the kinetic energy of the neutrons. Some of the latter are immediate (so-called prompt neutrons), but a small proportion (0.7% for U-235, 0.2% for Pu-239) is delayed, as these are associated with the radioactive decay of certain fission products. The longest delayed neutron group has a half-life of about 56 seconds.
The delayed neutron release is the crucial factor enabling a chain reacting system (or reactor) to be controllable and to be able to be held precisely critical. At criticality the chain reacting system is exactly in balance, such that the number of neutrons produced in fissions remains constant. This number of neutrons may be completely accounted for by the sum of those causing further fissions, those otherwise absorbed, and those leaking out of the system. Under these circumstances the power generated by the system remains constant. To raise or lower the power, the balance must be changed (using the control system) so that the number of neutrons present (and hence the rate of power generation) is either reduced or increased. The control system is used to restore the balance when the desired new power level is attained.
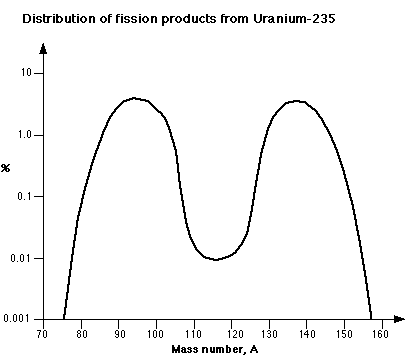
The number of neutrons and the specific fission products from any fission event are governed by statistical probability, in that the precise break up of a single nucleus cannot be predicted. However, conservation laws require the total number of nucleons and the total energy to be conserved. The fission reaction in U-235 produces fission products such as Ba, Kr, Sr, Cs, I and Xe with atomic masses distributed around 95 and 135. Examples may be given of typical reaction products, such as:
U-235 + n ===> Ba-144 + Kr-90 + 2n + energy
U-235 + n ===> Ba-141 + Kr-92 + 3n + 170 MeV
U-235 + n ===> Zr-94 + La-139 + 3n + 197 MeV
In such an equation, the number of nucleons (protons + neutrons) is conserved, e.g. 235 + 1 = 141 + 92 + 3, but a small loss in atomic mass may be shown to be equivalent to the energy released. Both the barium and krypton isotopes subsequently decay and form more stable isotopes of neodymium and yttrium, with the emission of several electrons from the nucleus (beta decays). It is the beta decays, with some associated gamma rays, which make the fission products highly radioactive. This radioactivity (by definition!) decreases with time.
The total energy released in fission varies with the precise break up, but averages about 200 MeV* for U-235 or 3.2 x 10-11 joule. That from Pu-239 is about 210 MeV* per fission. (This contrasts with 4 eV or 6.5 x 10-19 J per molecule of carbon dioxide released in the combustion of carbon in fossil fuels.)
* these are total available energy release figures, consisting of kinetic energy values (Ek) of the fission fragments plus neutron, gamma and delayed energy releases which add about 30 MeV.
About 6% of the heat generated in the reactor core originates from radioactive decay of fission products and transuranic elements formed by neutron capture, mostly the former. This must be allowed for when the reactor is shut down, since heat generation continues after fission stops. It is this decay which makes spent fuel initially generate heat and hence need cooling. Even after one year, typical spent fuel generates about 10 kW of decay heat per tonne, decreasing to about 1 kW/t after ten years.
Storage pond for spent fuel at UK reprocessing plant
Since U-238 is the major proportion of the fuel element material in a thermal reactor, capture of neutrons by U-238 and the creation of U-239 is an important process.
As already noted, Pu-239 is fissile in the same way as U-235, i.e. with thermal neutrons. It is the other main source of energy in any nuclear reactor and typically contributes about one third of the energy output. The masses of its fission products are distributed around 100 and 135 atomic mass units.
The main transuranic constituents of spent fuel are isotopes of plutonium, neptunium and americium. These are alpha-emitters and have long half-lives, decaying on a similar time scale to the uranium isotopes. They are the reason that spent fuel needs secure disposal beyond the few thousand years or so which might be necessary for the decay of fission products alone.
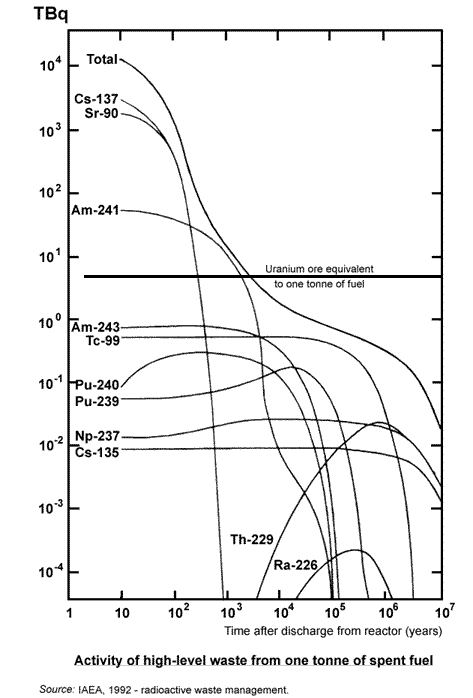
Apart from transuranic elements in the reactor fuel, activation products are formed wherever neutrons impact on any other material. Activation products in a reactor (and particularly its steel components exposed to neutrons) range from tritium (H-3) and carbon-14 to cobalt-60, iron-55 and nickel-63. The latter four radioisotopes create difficulties during eventual demolition of the reactor, and affect the extent to which materials can be recycled.
Such reactors are more efficient at converting fertile material than ordinary thermal reactors because of the arrangement of fissile and fertile materials, and there is some advantage from the fact that Pu-239 yields more neutrons per fission than U-235. Although both yield more neutrons per fission when split by fast rather than slow neutrons, this is incidental since the fission cross sections are much smaller at high neutron energies. Fast neutron reactors may be designed as breeders to yield more fissile material than they consume or to be plutonium burners to dispose of excess plutonium. A plutonium burner would be designed without a breeding blanket, simply with a core optimised for plutonium fuel.
For instance, the Fast Breeder Reactor was originally conceived to extend the world's uranium resources, and could do this by a factor of about 60. Although several countries ran extensive fast breeder reactor development programs, major technical and materials problems were encountered. To the extent that these programs permitted, it was not established that any of the designs would have been commercially competitive with existing light water reactors. An important aspect of fast reactor economics lies in the value of the plutonium fuel which is bred; unless this shows an advantage relative to contemporary costs for uranium, there would be little benefit from the use of this type of reactor. This point has been driven home today by the continuing abundance of uranium in geological resources and its low price. Nevertheless, there is international concern over the disposal of ex-military plutonium, and there are proposals to use fast reactors for this purpose. In both respects the technology is important to long term considerations of world energy sustainability.
The ability to control the chain reaction is entirely due to the presence of the small proportion of delayed neutrons arising from fission. Without these, any change in the critical balance of the chain reaction would lead to a virtually instantaneous and uncontrollable rise or fall in the neutron population. It is also relevant to note that safe design and operation of a reactor sets very strict limits on the extent to which departures from criticality are permitted. These limits are built in to the overall design.

Sizewell B nuclear power plant, UK
Neutrons released in fission are initially fast (velocity about 109 cm/sec, or energy above 1 MeV), but fission in U-235 is most readily caused by slow neutrons (velocity about 105 cm/sec, or energy about 0.02 eV). A moderator material comprising light atoms thus surrounds the fuel rods in a reactor. Without absorbing too many, it must slow down the neutrons in elastic collisions (compare it with collisions between billiard balls on an atomic scale). In a reactor using natural (unenriched) uranium the only suitable moderators are graphite and heavy water (these have low levels of unwanted neutron absorption). With enriched uranium (ie. increased concentration of U-235 - see Appendix), ordinary (light) water may be used as moderator. (Water is also commonly used as a coolant, to remove the heat and generate steam.)
Other features may be used in different reactor types to control the chain reaction. For instance, a small amount of boron may be added to the cooling water and its concentration reduced progressively as other neutron absorbers build up in the fuel elements. (For emergency situations, provision may be made for rapidly adding an excessive quantity of boron to the water.)
Commercial power reactors are usually designed to have negative temperature and void coefficients. The significance of this is that if the temperature should rise beyond its normal operating level, or if boiling should occur beyond an acceptable level, the balance of the chain reaction is affected so as to reduce the rate of fission and hence reduce the temperature. One mechanism involved is the Doppler effect, whereby U-238 absorbs more neutrons as the temperature rises, thereby pushing the neutron balance towards subcritical. Another mechanism, in light water reactors, is that the formation of steam within the water moderator will reduce its density and hence its moderating effect, and this again will tilt the neutron balance towards subcritical.
While fuel is in use in the reactor, it is gradually accumulating fission products and transuranic elements which cause additional neutron absorption. The control system has to be adjusted to compensate for the increased absorption. When the fuel has been in the reactor for three years or so, this build-up in absorption, along with the metallurgical changes induced by the constant bombardment of the fuel materials, dictates that the fuel should be replaced. This effectively limits the burn-up to about half of the fissile material, and the fuel assemblies must then be removed and replaced with fresh fuel.
In naval reactors used for propulsion, where frequent fuel changes are inconvenient, the fuel is enriched to higher levels initially and burnable poisons - neutron absorbers - are incorporated. Hence as the fission products and transuranic elements accumulate, the 'poison' is depleted and the two effects tend to cancel one another out. As engineers explore ways to increase the burn-up of commercial reactor fuel, burnable poisons such as gadolinium may be used along with increasing enrichment towards 5% U-235 (as is already happening).
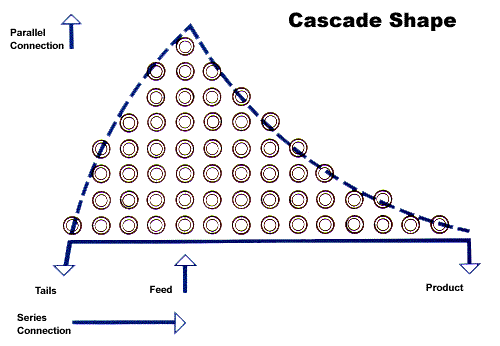
The two main enrichment (or isotope separation) processes are diffusion (gas diffusing under pressure through a membrane containing microscopic pores) and centrifugation. In each case, a very small amount of isotope separation takes place in one pass through the process. Hence repeated separations are undertaken in successive stages, arranged in a cascade. The product from each stage becomes feed for the next stage above, and the depleted material is added to the feed for the next stage below. The stages above the initial feed point thus become the enriching section and those below are the stripping section. Each stage thus has a double feed (product from below and depleted from above). Ultimately, the enriched product is about one sixth or one seventh the amount of depleted material, so that the product end of the cascade tends to have more stages. The depleted material, drawn off at the bottom of the stripping section, is commonly called tails and the residual U-235 concentration in the tails is the tails assay.
Source: Urenco
The separating power of the cascade, or of each stage, is described in terms of flow capacity and enriching ability, using the separative work unit (SWU) to quantify it. This is dimensionally a mass unit, though it indicates energy (for a particular plant, energy consumption may be described in kWh per SWU). Since feed or product quantities are measured in tonnes or kilograms, SWUs are also described similarly.
For instance, to produce one kilogram of uranium enriched to 3.5% U235 requires 4.3 SWU if the plant is operated at a tails assay 0.30%, or 4.8 SWU if the tails assay is 0.25% (thereby requiring only 7.0 kg instead of 7.8 kg of natural U feed). Here, and in the following paragraph, kg SWU units are implied.
About 100,000 to 120,000 SWU is required to enrich the annual fuel loading for a typical 1000 MWe light water reactor. Enrichment costs are related to electrical energy used. The gaseous diffusion process consumes up to 2,400 kWh (8,600 MJ) per SWU, while gas centrifuge plants require only about 50 kWh/SWU (180 MJ). Despite this, competition between the commercial enrichment plants compels comparable prices to be charged, and the relative capital costs and ages of the enrichment plants may compensate the large difference in the electric power components.
The diffusion process relies on a difference in average velocity of the two types of UF6 molecules to drive the lighter ones more readily through holes in the membranes. Each stage consists of a compressor, a diffuser and a heat exchanger to remove the heat of compression. The enriched UF6 product is withdrawn from one end of the cascade and the depleted UF6 is removed at the other end. The gas must be processed through some 1400 stages to obtain a product with a concentration of 3% to 4% U-235. Diffusion plants typically have a small amount of separation through one stage (hence the large number of stages) but are capable of handling large volumes of gas.

The large Tricastin enrichment plant in France (beyond cooling towers)
The four nuclear reactors in the foreground provide over 3000 MWe power for it.
Centrifuge enrichment relies on the simple mass difference of the molecules coupled with the square of the peripheral velocity in a rapidly rotating cylinder (the centrifuge rotor). Countercurrent movement of gas within the rotor, proportional to its height, enhances this effect. The gas is fed into a series of evacuated cylinders, each containing a rotor about one to two metres long and 15-20 cm diameter. When the rotors are spun rapidly, the heavier molecules with U-238 increase in concentration towards the cylinder's outer edge, leaving a corresponding increase in concentration of molecules with U-235 near the centre. The countercurrent flow enables enriched product to be drawn off axially.
To obtain efficient separation of the two isotopes, centrifuges rotate at very high speeds, typically 50,000 to 70,000 rpm, with the outer wall of the spinning cylinder moving at between 400 and 500 metres per second, to give a million times the acceleration of gravity. There are considerable materials and engineering challenges in producing such equipment.

A bank of centrifuges at a European plant
Although the volume capacity of a single centrifuge is much smaller than that of a single diffusion unit, its ability to separate isotopes is much greater. Centrifuge stages normally consist of a large number of centrifuges in parallel. Such stages are then arranged in cascade similarly to those for diffusion. In the centrifuge process, the number of stages may only be 10 to 20, instead of the thousand or more required for diffusion.
Laser isotope separation processes have been a focus of interest for some time. They promise lower energy inputs, lower capital costs and lower tails assays, hence significant economic advantages. None of these processes is yet ready for commercial use, and the US AVLIS process, into which billions of dollars had been invested and which was thought to be in an advanced stage of development, was cancelled in 1999.
Laser processes utilise the very precise beam frequencies characteristic of lasers. Such frequencies are equivalent to defined energies. The interaction of the laser beam with gas or vapour enables it to exploit the excitation or ionisation of isotope-specific atoms in the vapour. It may then be possible to separate molecules containing a desired isotope by utilising a second physical process applicable only to the excited or ionised molecule. For instance a tuned laser of very specific energy might convert UF6 molecules containing U-235 atoms to solid UF5, by breaking the molecular bond holding the sixth fluorine atom. This then enables the UF5 to be separated from the unaffected UF6 molecules containing U-238 atoms, hence achieving a separation of isotopes.
Laser separation processes may use either atomic or molecular gases or vapours. AVLIS is an atomic process. A molecular laser separation process, SILEX, utilising uranium in the form of UF6, was invented in Australia, and is currently under development in collaboration with a US licensee.
Back To Home