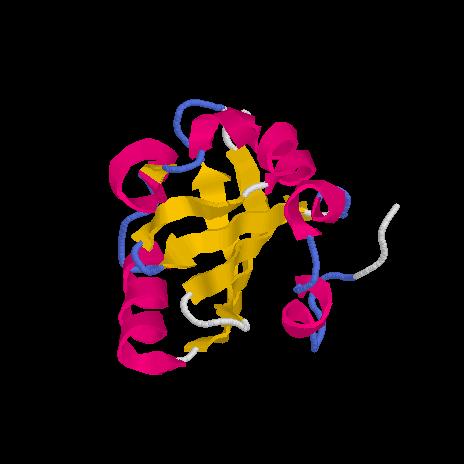
Before it was actually observed in nature, the structure of the alpha helix was predicted by Linus Pauling based on the planar atomic structure of the peptide bond and the optimal hydrogen-bonding geometry this structure permits. Many proteins contain one or more stretches of amino acids that take on a characteristic structure in 3-D space, the most common being the beta conformation and the alpha helix .
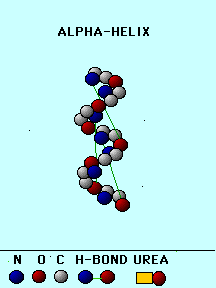
An alpha-helix is formed from a connected stretch of amino acids. Its main characteristics are:
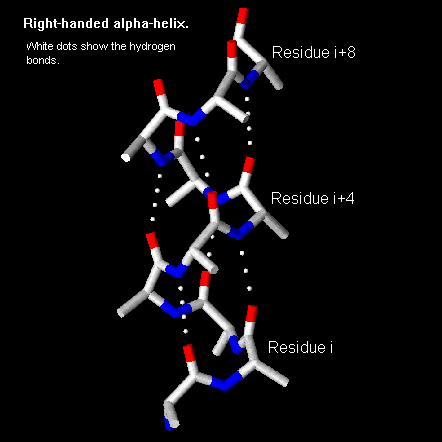
An average alpha helix is 10 residues long, although they can range between 4-40 residues in length. All residues have similar phi and psi angles. The standard angles for a right-handed alpha-helix are
f =-57° , y = -47° , w =180° . The w stays near 180° because the peptide bond is planar due to a partial double bond being formed.Certain amino acids are preferred in an alpha helix, such as Ala, Glu, Leu, and Met. Pro, Gly, Tyr, and Ser have a small tendency to do the same. Proline cannot actually fit into a helix, and introduces a kink into the structure.
The side-chains point out of the helix. The overall hydrophobicity of a helix is determined by the side chains. If the side chains are hydrophobic, the helix is usually buried somewhere within the proteins core, away from the solvent. If the side chains are polar or charged (hydrophilic), the helix is probably exposed to the solvent. Some helices have a dual character (one side hydrophobic, the other side hydrophilic), and may be found at the protein's surface.
FAVORITE LINKS:
Excellent pictures of alpha helix
Great overview of alpha helix protein structure
Structural aspects of proteins
Awesome graphics of DNA and proteins
Pictures of other Helical Structures
My Favorite Links
Back to Home Page
Overview of Secondary Structures
Beta Sheet Structures